1. Important Characteristics & Limitations
Key Points for JEE/NEET
- Substrate Suitability: Excellent for ketones containing phenolic or carboxylic acid groups, as these remain unaffected by the acidic conditions.
- Limitations: The reaction fails with acid-sensitive substrates (e.g., acetals, ketals, or molecules with easily dehydrated -OH groups) and high molecular weight substrates.
- Conjugated Systems: α,β-unsaturated ketones undergo reduction of both the olefinic (double bond) and the carbonyl groups.
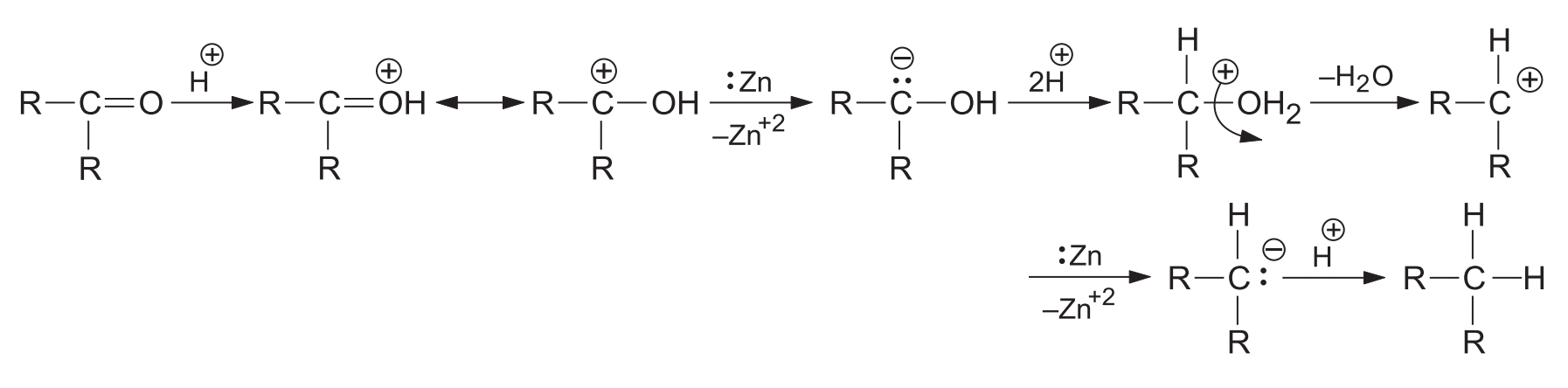
2. Mechanism of Clemmensen Reduction
While various mechanisms have been proposed, the most accepted one was suggested by Tadaaki Nakabayashi. It assumes that under acidic conditions, the carbonyl group gets protonated, and electrons are directly transferred from the zinc metal.
Note: A mechanism suggesting the intermediate formation of an alcohol was rejected because the reagent fails to reduce most alcohols to hydrocarbons.
3. Exceptions and Special Cases (Advanced Level)
Anomalous Reactions
Certain aldehydes and ketones do not give the normal reduction products:
- α-hydroxy ketones: Give either ketones (through hydrogenolysis of the -OH group) or olefins.
- 1,3-diketones: Give exclusively monoketones accompanied by carbon skeleton rearrangement.
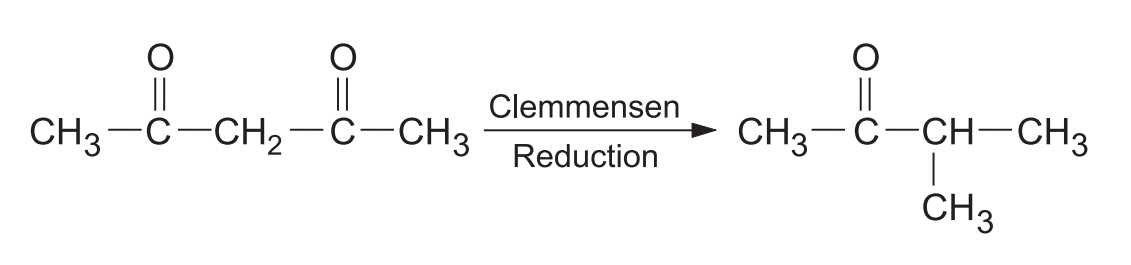
- Cyclic 1,3-diketones: Give a fully reduced product along with a monoketone resulting from ring contraction.
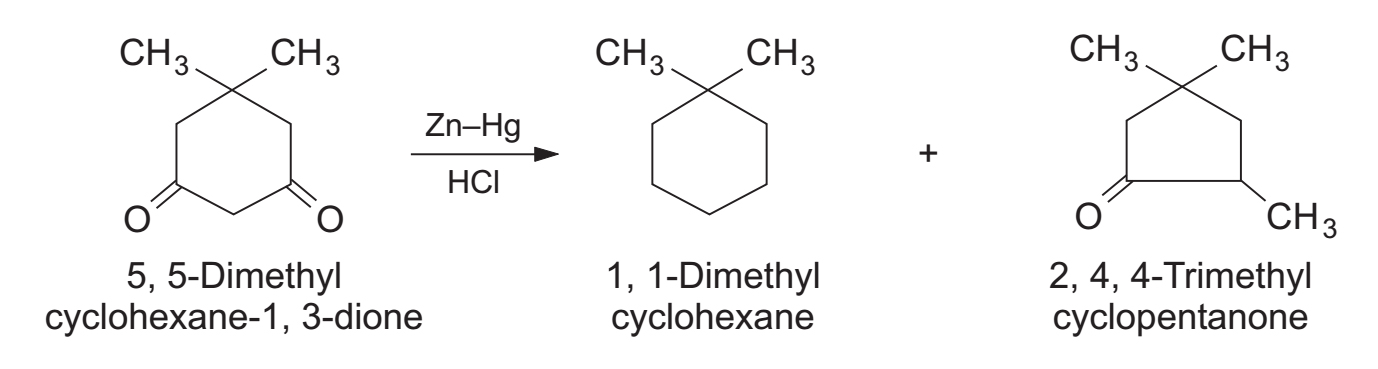
4. Synthetic Applications
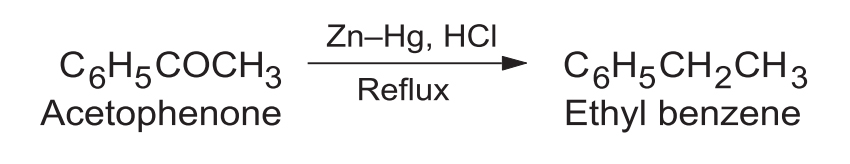
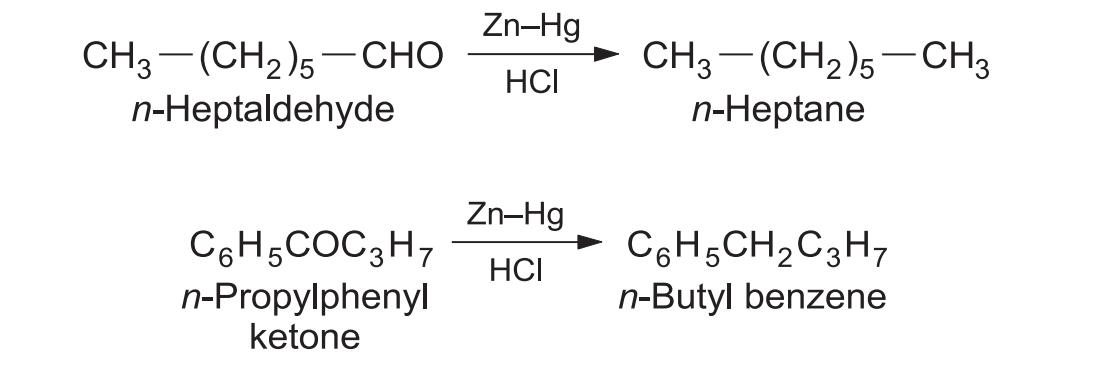
1. Aliphatic & Aromatic Carbonyls
Highly useful for introducing straight-chain alkyl groups into aromatic rings (Friedel-Crafts acylation followed by Clemmensen reduction) to avoid carbocation rearrangement.
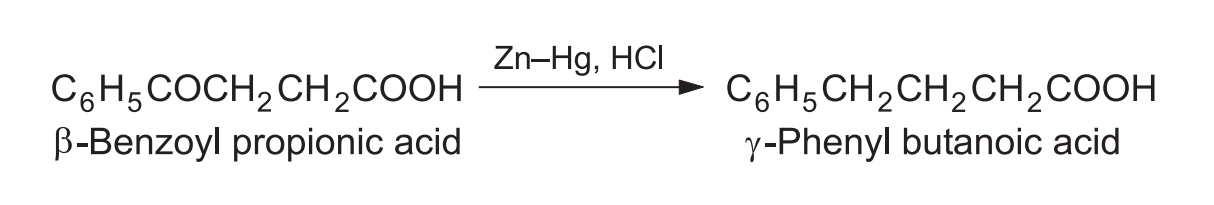
2. Reduction of Keto Acids
Works well for
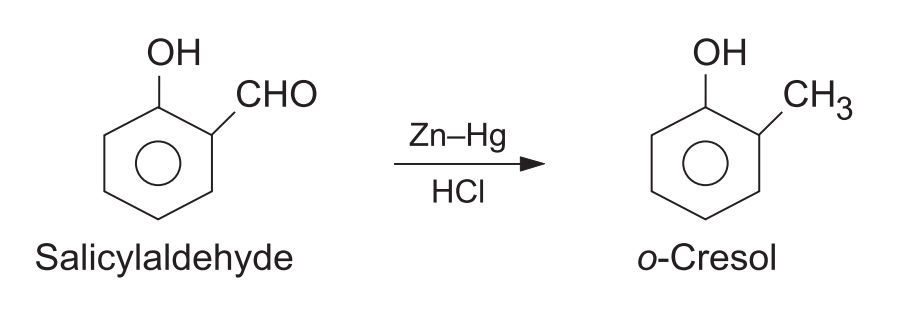
3. Phenolic Carbonyl Compounds
The phenolic -OH group is unaffected by the acidic conditions.
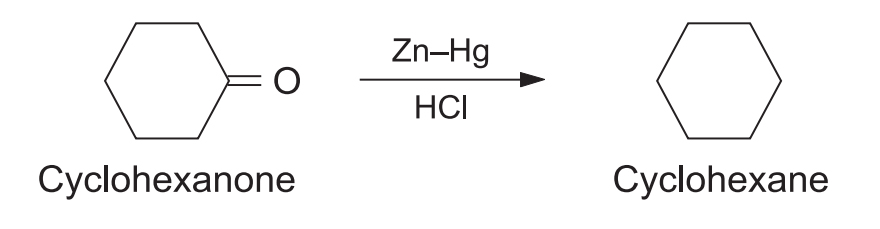
4. Synthesis of Cycloparaffins
Used to close rings or reduce cyclic ketones.