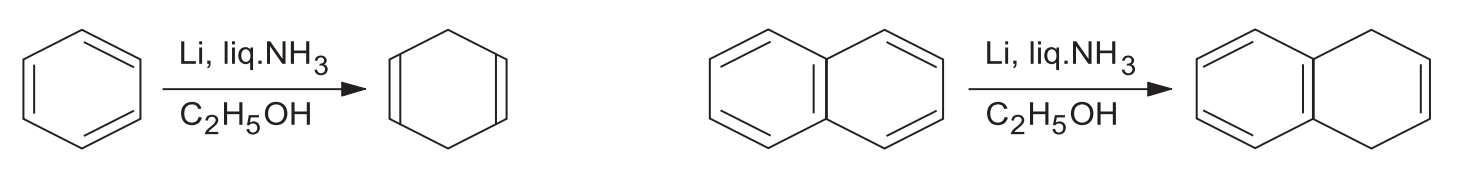
1. Mechanism of Birch Reduction
The mechanism proceeds via a series of single-electron transfers (SET) and protonations:
- Electron Transfer: The alkali metal transfers one electron to the benzene ring, producing a resonance-stabilized anion radical (Ia–Ic).
- First Protonation: The anion radical accepts a proton from the alcohol to form a neutral radical (II).
- Second Electron Transfer: The addition of another electron from the metal to the radical produces a carbanion (III).
- Second Protonation: The anion (III) subsequently takes up another proton from the alcohol to yield the final 1,4-dihydro product.
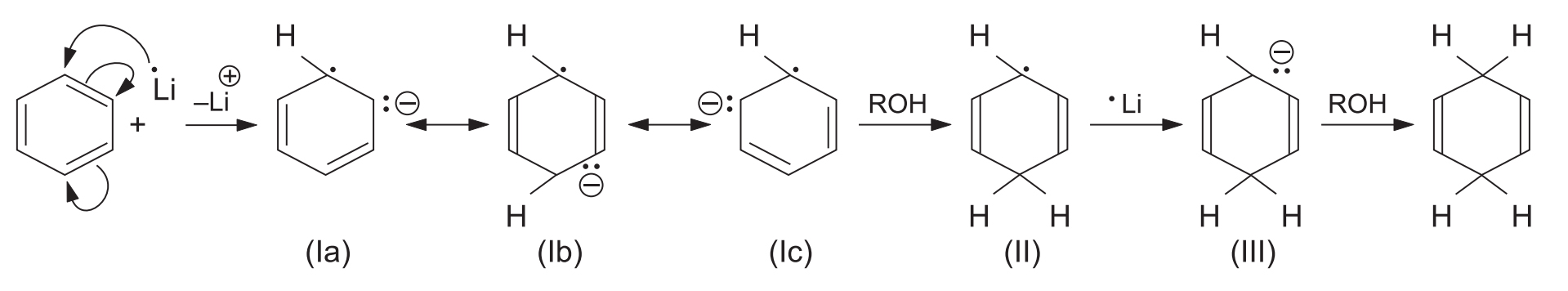
Why 1,4-addition? The repulsion between the anionic and radical centres is minimum in structure (Ib). Therefore, it adds a proton to give (II), ensuring a 1,4-dihydro product rather than a 1,2-dihydro product.
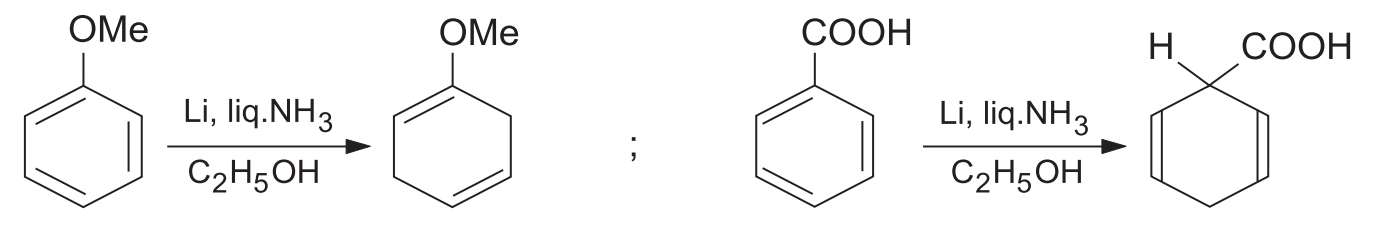
2. Regioselectivity: Effect of Substituents (Crucial for JEE/NEET)
The presence of substituents on the benzene ring drastically affects the outcome of the reduction.
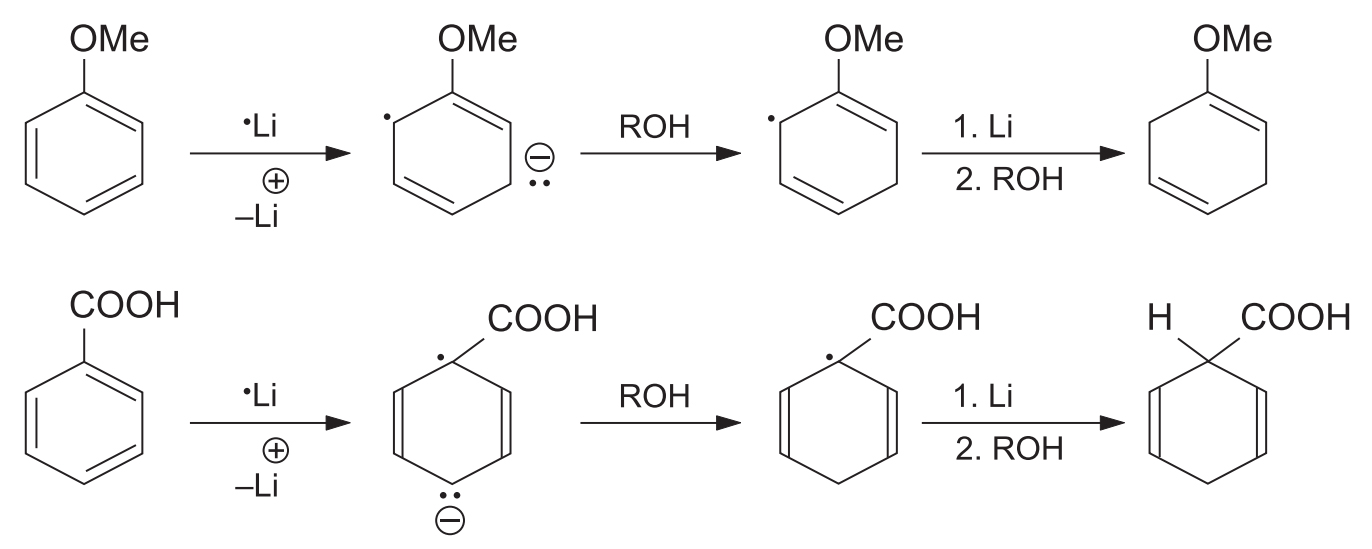
- Electron-Withdrawing Groups (EWG): Groups like -COOH or -NO2 make the ring more electron-accepting, facilitating the reaction. The electron-withdrawing group remains on the saturated sp3 carbon in the product (i.e., reduction occurs at the ipso and para positions).
- Electron-Donating Groups (EDG): Groups like -OCH3 or -CH3 have a retarding effect. The electron-donating group remains on the unsaturated sp2 carbon in the product (i.e., reduction occurs at the ortho and meta positions).
This regioselectivity is explained by molecular orbital calculations, which show that in the anion-radical:
- With an EDG, electron density is highest at the ortho or meta positions.
- With an EWG, electron density is highest at the para position.
3. Exceptions and Special Conditions
High Temperature Variation
At higher temperatures (50–120 °C), ammonia itself becomes the proton source, and alcohol is not needed.
The amide ion (NH2-) formed is a strong base and isomerizes the unconjugated 1,4-dihydro product into a conjugated 1,2-dihydro product.
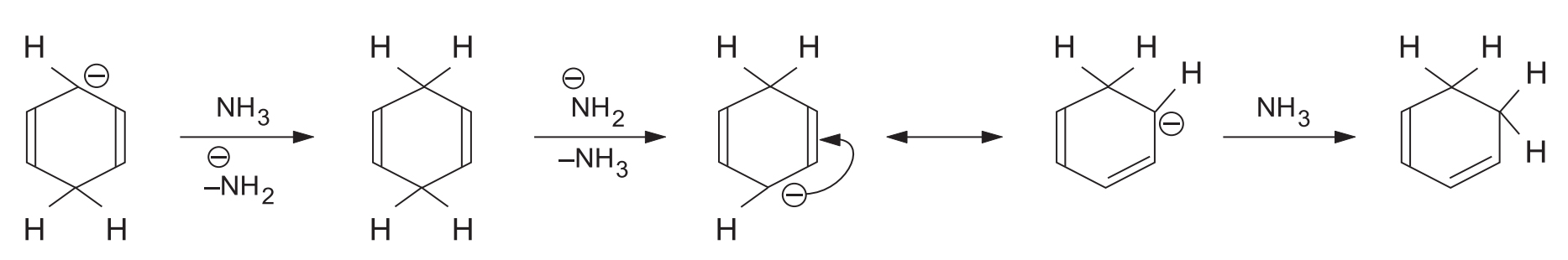
Because the 1,2-dihydro product has a conjugated double bond, it undergoes further reduction to form a tetrahydro derivative.
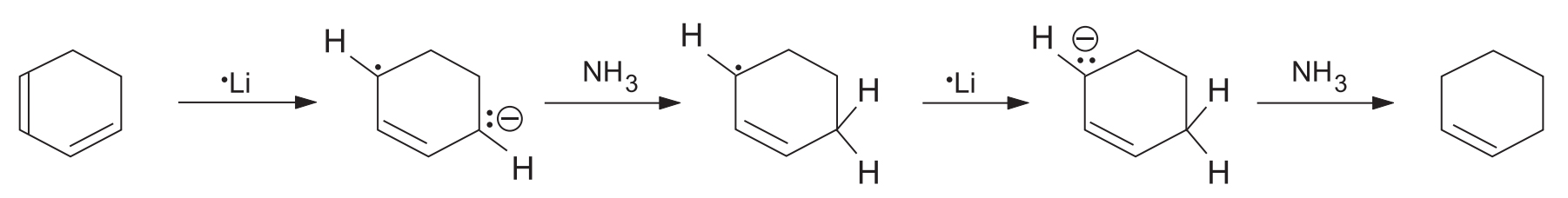
Important Limitations
- Isolated Double Bonds: Olefinic bonds like the one in cyclohexene are unaffected by the Birch reagent.
- Phenols: Phenols are generally not reduced by this method due to the formation of phenoxide ions in the basic medium, which repel the incoming electrons.