1. Reagents and Substrates
Common Catalysts
The reaction is catalyzed by acidic reagents that can convert the hydroxyl group into a better leaving group. Common reagents include:
- Protic acids: H2SO4, Polyphosphoric acid (PPA).
- Lewis acids / Dehydrating agents: P2O5, PCl5, SOCl2, SO3.
- Sulfonyl chlorides: C6H5SO2Cl (Benzenesulfonyl chloride).
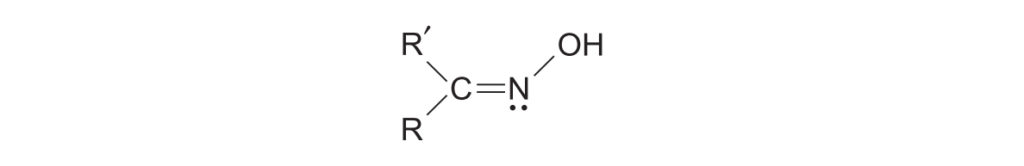
Note on Aldoximes: While ketoximes rearrange smoothly, aldoximes generally do not undergo this reaction as a general rule, though some may rearrange in the presence of PPA.
2. Stereochemistry: The "Anti" Migration Rule (Crucial for JEE)
The most important feature of the Beckmann Rearrangement is that it is stereospecific.
- The migration of the group from carbon to electron-deficient nitrogen depends not on the general migrational aptitude (unlike the Baeyer-Villiger oxidation), but entirely upon the spatial orientation of the groups.
- The Rule: The migrating group is always the one that is anti (i.e., trans) to the hydroxyl (-OH) group on the oxime.
Proof of Stereochemistry
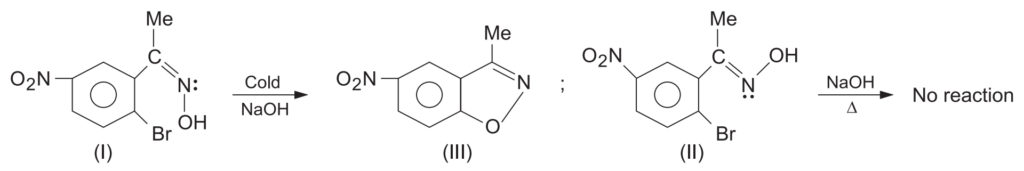
The classic proof of anti-migration comes from the rearrangement of the two isomeric oximes of 2-bromo-5-nitroacetophenone.
- Structural Assignment: When treated with cold NaOH, isomer (I) cyclized to an isooxazole derivative, meaning the -OH and -Br groups were close enough (cis/syn) to react. Therefore, the Methyl (Me) and -OH groups were anti to each other. Isomer (II) did not react, proving its -OH and -Br groups were far apart (Me and -OH are syn).
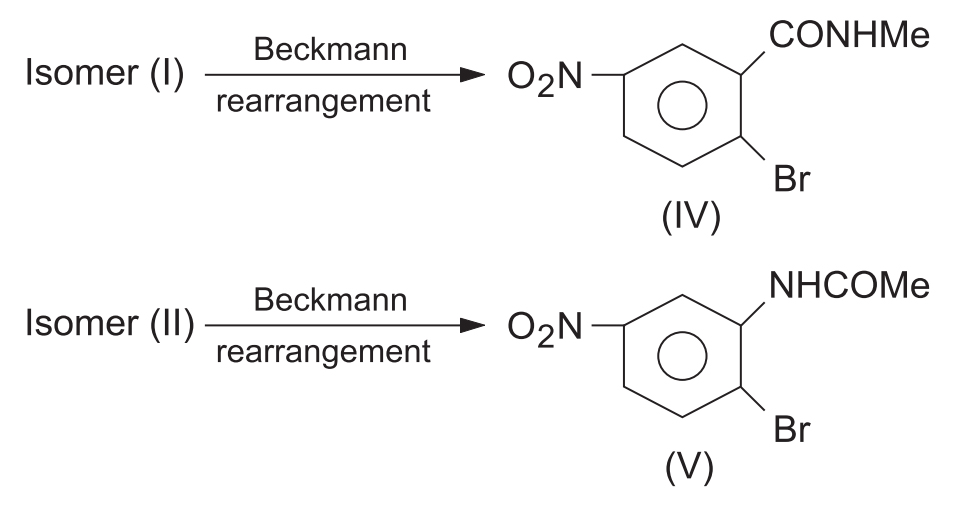
- Rearrangement Result: Upon undergoing the Beckmann rearrangement, isomer (I) gave a product indicating the migration of the Me group (which was anti to OH). Isomer (II) gave a product indicating the migration of the Aryl group (which was anti to OH).
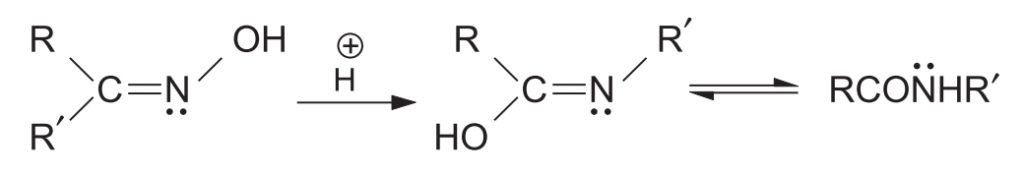
3. Mechanism of Beckmann Rearrangement
The mechanism proceeds via the formation of an electron-deficient nitrogen. The reaction is facilitated by heat, polar solvents, or an increase in acid strength.
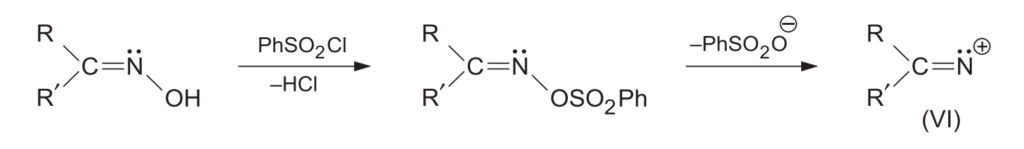
- Activation: In strong acids, the -OH group of the oxime is protonated. With other reagents (like PCl5 or PhSO2Cl), the -OH is converted into an ester-leaving group (e.g., OPCl4 or OSO2Ph).
- Migration and Departure: The leaving group (H2O or ester group) departs, and simultaneously, the group anti to it migrates to the electron-deficient nitrogen. This process is concerted or very rapid, ensuring the migrating group retains its configuration and does not become completely free.
- Hydration: A water molecule attacks the resulting carbocation.
- Tautomerization: Loss of a proton leads to an imidic acid intermediate, which tautomerizes to the final amide.
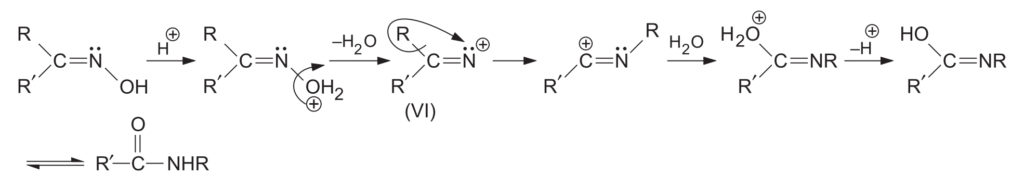
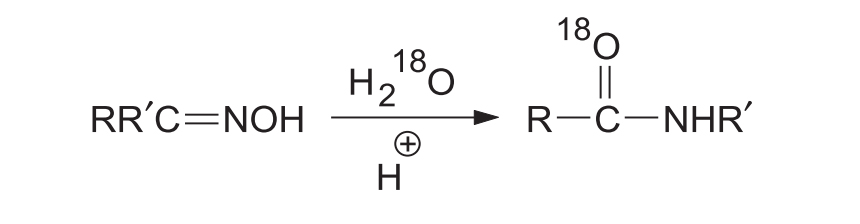
Isotope Labelling Proof: If the reaction is carried out in the presence of H218O, 18O is incorporated into the final product. This proves that the original -OH group leaves entirely and a direct interchange of the migrating group and OH does not occur.
4. Applications of Beckmann Rearrangement
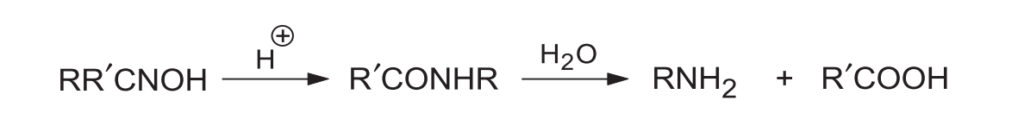
- 1. Determining Configuration of Ketoximes: By identifying the hydrolysis products of the resulting amide (RNH2 + R'COOH), one can determine which group migrated (and therefore, which group was anti to the -OH in the original oxime).
- 2. Synthesis of Isoquinoline: Used in advanced organic synthesis pathways to form fused heterocyclic rings.
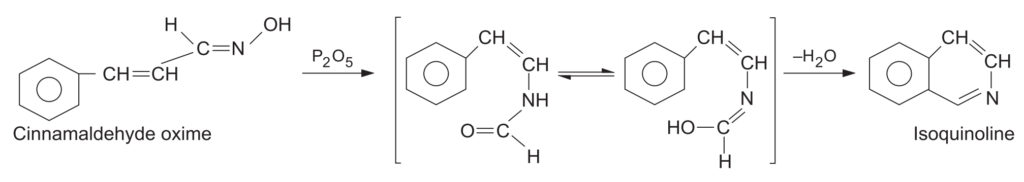
- 3. Synthesis of Lactams: Alicyclic ketones (cyclic ketones) of all ring sizes undergo Beckmann rearrangement of their oximes to yield cyclic amides known as lactams. *(A classic JEE example is the conversion of cyclohexanone oxime to caprolactam, the monomer for Nylon-6).*
- 4. Synthesis of Paracetamol: Used industrially to integrate the amino and phenolic functionalities. This involves converting a ketone to a ketoxime using hydroxylamine, followed by rearrangement.
