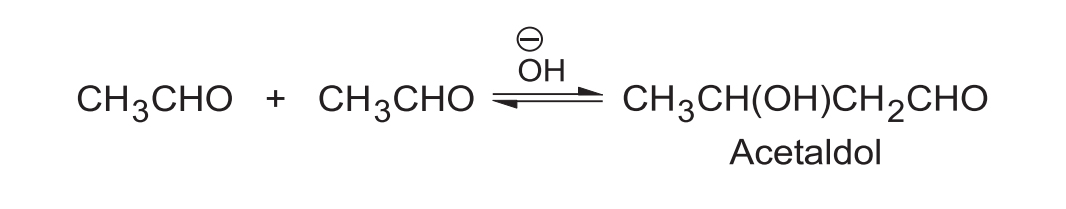
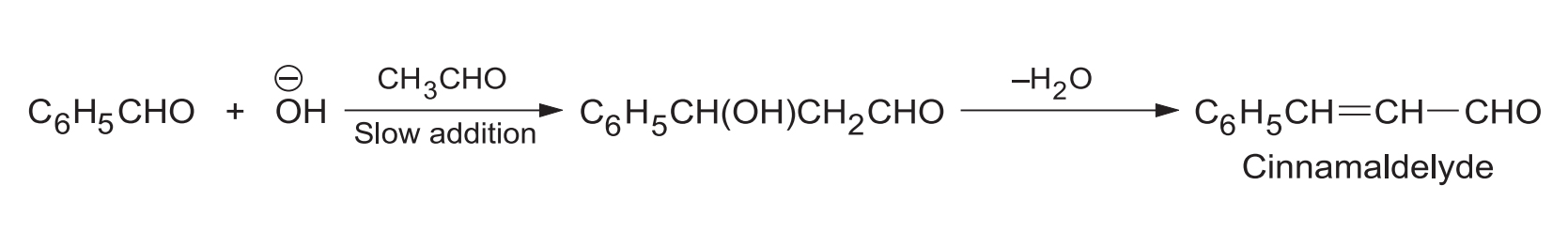Aldehydes having -hydrogen(s) undergo self-condensation on warming with dilute or mild base to give -hydroxy aldehydes, called aldols (aldehyde + alcohol). This reaction is known as aldol condensation. A typical example is the reaction of acetaldehyde with base under mild condition.
Various basic reagents such as dilute sodium hydroxide, aqueous alkali carbonate, alkali metal alkoxides, etc., may be used. The reaction is not favourable for ketones. For IIT JEE and NEET, mastering this reaction is critical as it frequently appears in multi-step synthesis problems.
1. Scope and Types of Aldol Condensation
Aldol condensation has broad scope and can occur between:
- (i) two identical or different aldehydes
- (ii) two identical or different ketones
- (iii) an aldehyde and a ketone
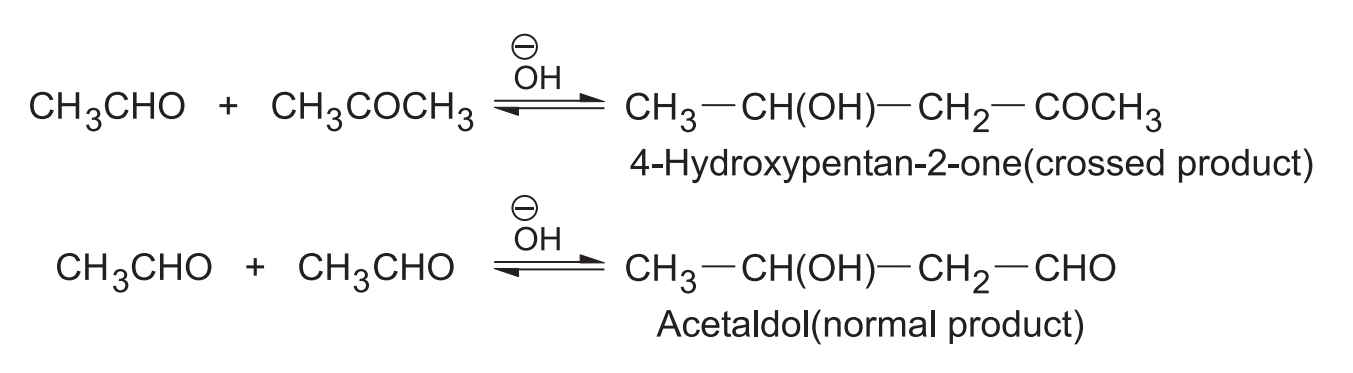
When the condensation is between two different carbonyl compounds, it is called crossed aldol condensation.
2. Mechanism of Aldol Condensation
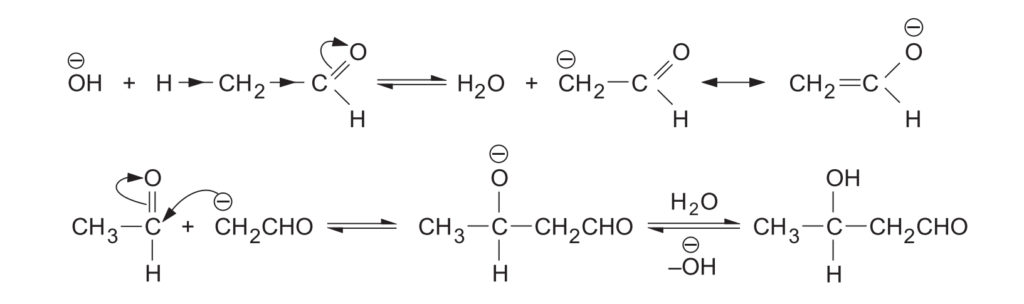
The overall reaction is an addition of an enolate anion to the carbonyl double bond.
- Step 1: The first step involves the formation of a resonance-stabilized enolate anion by the removal of an -hydrogen from the aldehyde by the base.
- Step 2: In the second step the enolate anion attacks the carbonyl carbon of the second molecule of the aldehyde to form an alkoxide ion.
- Step 3: The latter then takes up a proton from the solvent to yield aldol in the third step.
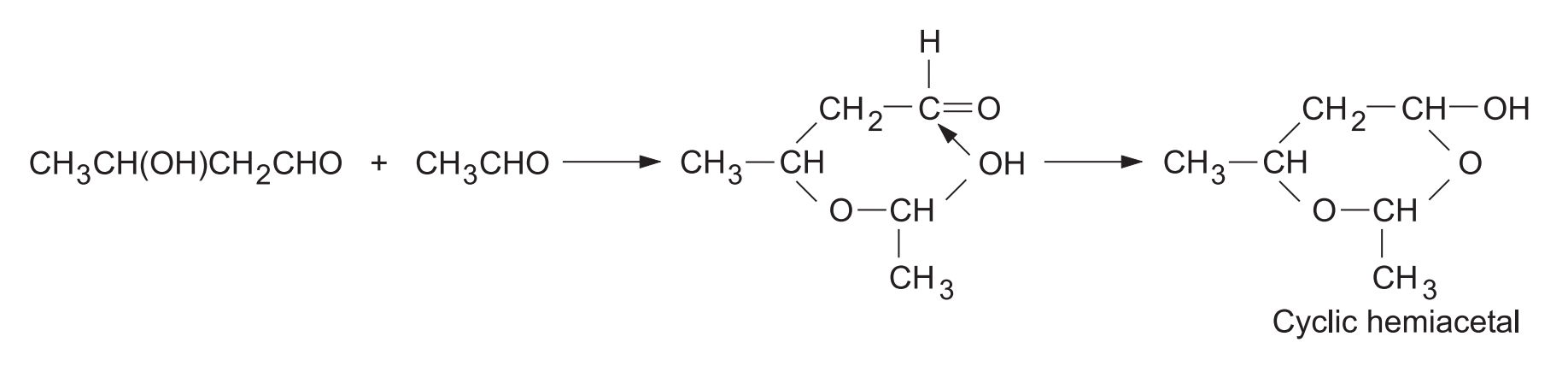
Usually aldol as such is not isolated; e.g., acetaldol is isolated as a cyclic hemiacetal. Aldol is isolated under reasonable mild condition, i.e., using aqueous as base.
Why are Ketones less reactive?
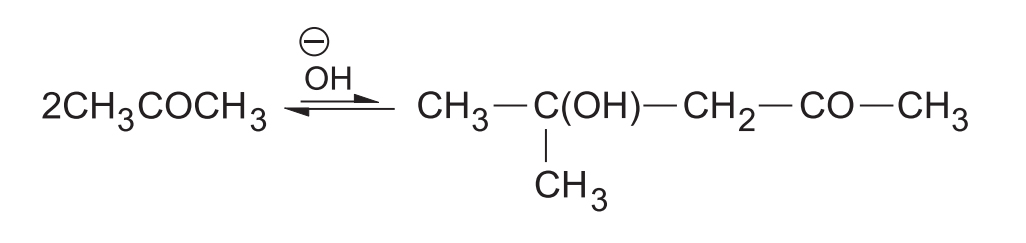
The reaction between two ketones is not very successful; the equilibrium is not favourable and lies far to the left.
- This is because the carbonyl carbon of ketone is less positive (due to +I effect) and more sterically hindered relative to aldehydes.
- This reduces the nucleophilic attack on the carbonyl carbon.
- However, it is possible to prepare diacetone alcohol in reasonable good yield by boiling acetone with solid in a specially devised apparatus.
3. Salient Features of Aldol Condensation
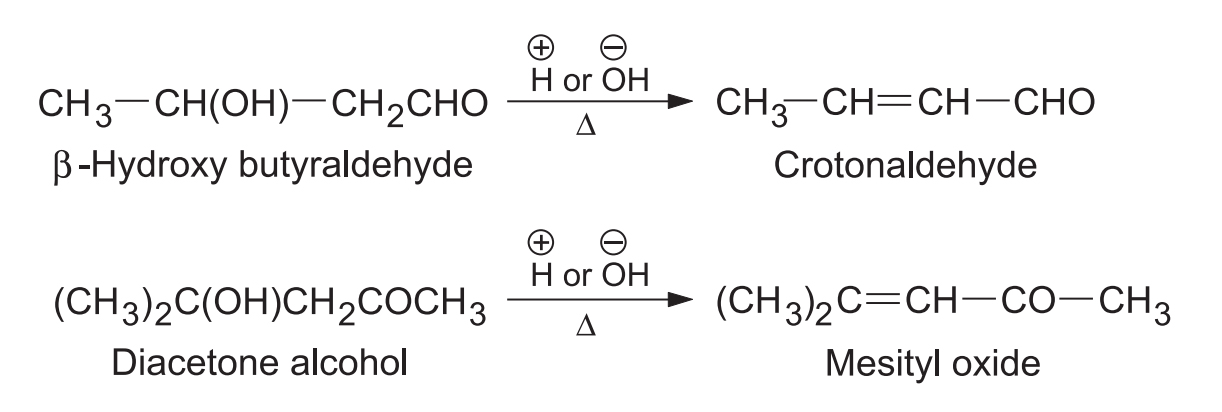
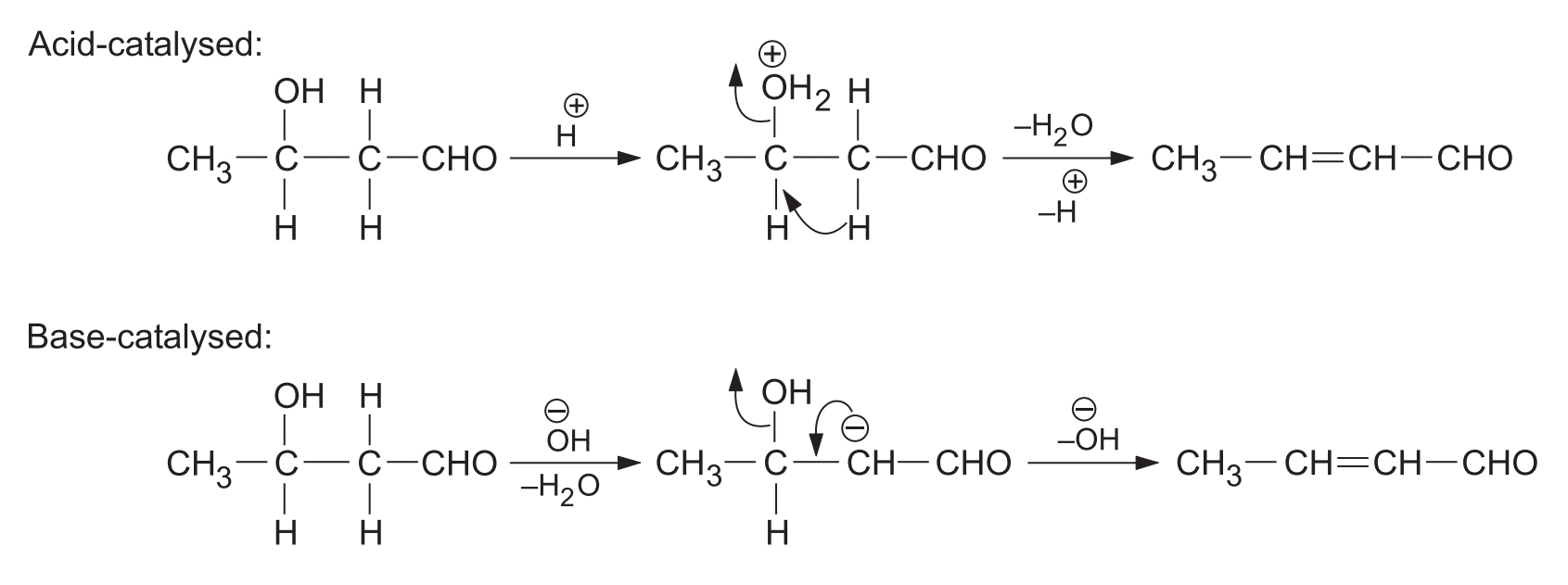
- (a) Dehydration: Aldols are easily dehydrated to -unsaturated compounds on heating alone or with acid or base.
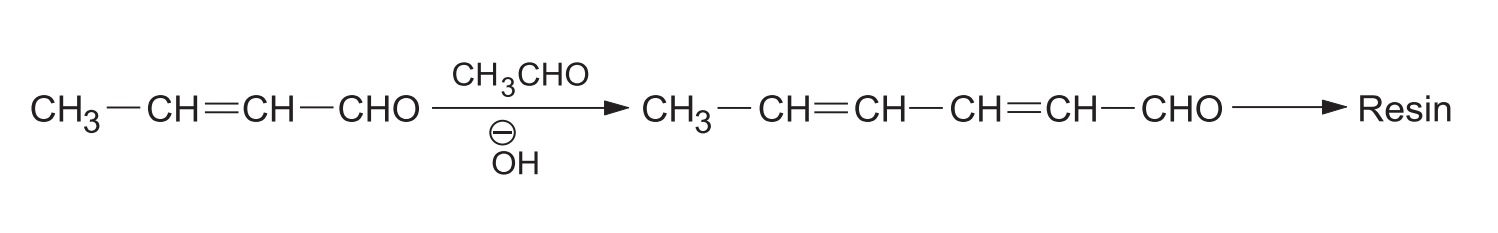
- (b) Resinification: When aldol condensation is carried out in the presence of strong alkali, repeated condensation and dehydration results in the formation of resins.
- (c) The condensation is promoted by –I effect and reduced by +I effect on the carbonyl carbon.
- (d) The reaction equilibrium is favourable for aldehydes but much less favourable for ketones.
4. Crossed Aldol Condensation
(i) Between two different aldehydes
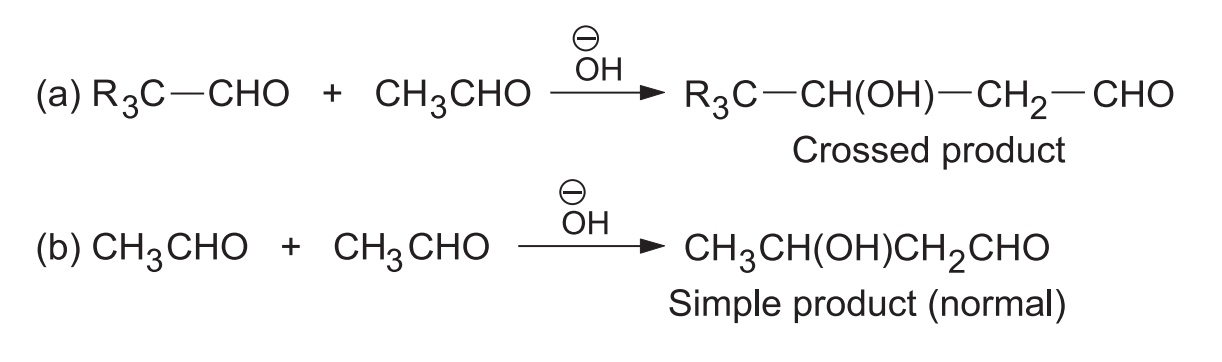
When both the aldehydes have -hydrogen(s) both can form carbanions and also can act as carbanion acceptors, forming a mixture of four products which has little synthetic use.
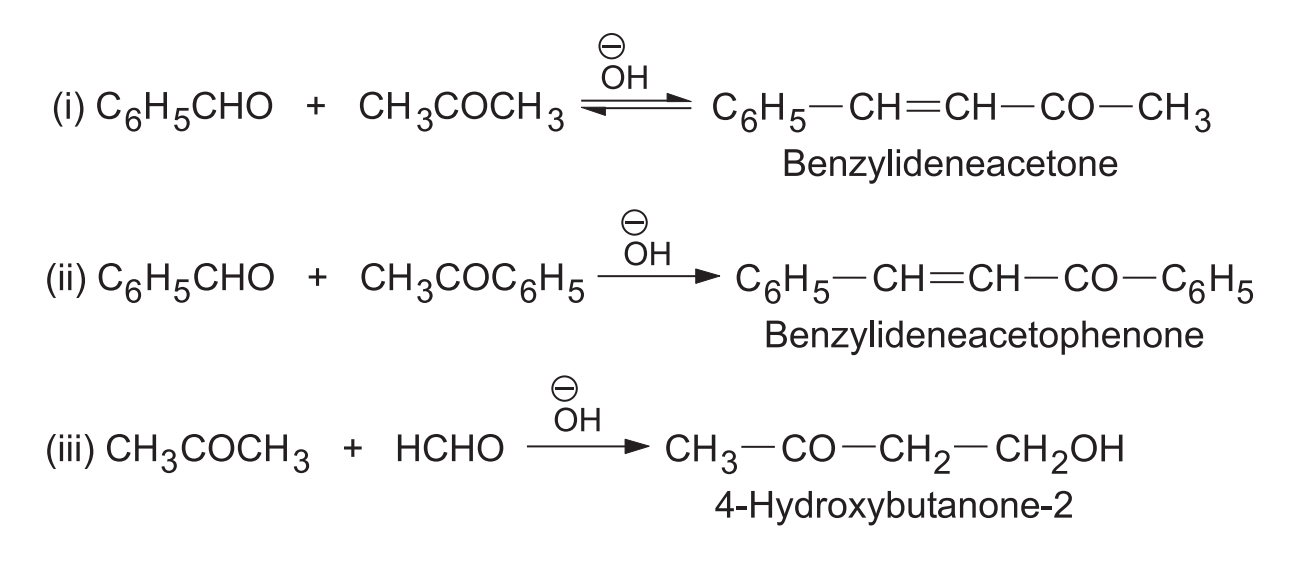
If one of the aldehydes has no -hydrogen then it can act only as a carbanion acceptor. In such case two products are formed.
A good yield of the crossed product is obtained by slowly adding the aldehyde having -hydrogen to a mixture of the aldehyde having no -hydrogen and the catalyst.
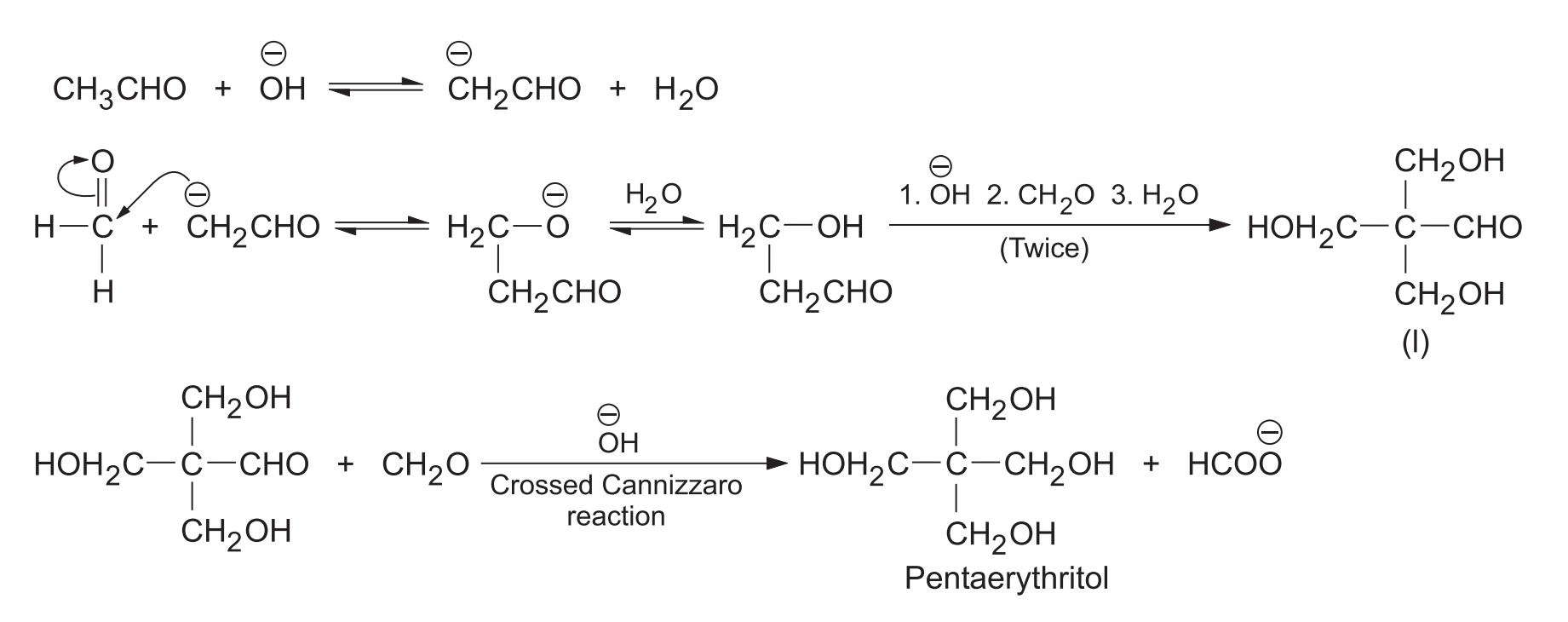
Formaldehyde having no -hydrogen is a reactive carbanion acceptor due to the absence of steric hindrance and +I effect. Hence, when acetaldehyde is treated with excess of formaldehyde in the presence of , crossed aldol condensation continues (three times) until trihydroxymethyl acetaldehyde is formed. The latter undergoes a crossed Cannizzaro reaction to form pentaerythritol.
(ii) Between two different ketones
Due to poor reactivity of carbonyl carbons (+I effect and crowding) of ketones, a poor yield is obtained and so it is rarely attempted.
(iii) Between an aldehyde and a ketone
(a) When an aldehyde and a ketone both having -hydrogens are condensed, two products are obtained. Aldehydes being more reactive than ketones act as carbanion acceptors and the ketones provide the carbanions.
(b) When the addition is between a ketone and an aldehyde with no -hydrogen, only one product is obtained. The ketone provides the carbanion and the aldehyde acts as the carbanion acceptor.
5. Dehydration of Aldols
Aldols can be easily dehydrated to -unsaturated compounds in the acidic or basic medium and sometimes on simple heating. The driving force for this dehydration is the formation of a highly stable extended conjugated system.
For molecules containing two carbonyl groups (diketones, dialdehydes, or keto-aldehydes), an Intramolecular Aldol Condensation can occur. Crucial Rule: The reaction will predominantly favor the formation of 5-membered or 6-membered rings due to minimum angle strain. Always form the enolate at a position that leads to a stable ring size when it attacks the other carbonyl group.
Practice Questions (JEE / NEET Level)
Q1: Which of the following compounds will NOT undergo Aldol condensation?
A) Acetaldehyde
B) Acetone
C) Benzaldehyde
D) Butanone
Answer: C) Benzaldehyde. It lacks -hydrogens, which are essential for the formation of the enolate ion.
Q2: A mixture of benzaldehyde and formaldehyde on heating with aqueous NaOH solution gives:
A) Benzyl alcohol and sodium formate
B) Sodium benzoate and methyl alcohol
C) Sodium benzoate and sodium formate
D) Benzyl alcohol and methyl alcohol
Answer: A) Benzyl alcohol and sodium formate. Since neither has -hydrogens, a Crossed Cannizzaro reaction occurs. Formaldehyde is oxidized to formate, and benzaldehyde is reduced to benzyl alcohol.
Q3: Why is the Aldol condensation equilibrium less favorable for ketones compared to aldehydes?
A) Ketones do not form enolates.
B) The carbonyl carbon of ketones is more sterically hindered and less electrophilic due to the +I effect of two alkyl groups.
C) Ketones undergo rapid Cannizzaro reaction.
D) The -hydrogens of ketones are not acidic.
Answer: B) The carbonyl carbon of ketones is more sterically hindered and less positive (due to +I effect), reducing nucleophilic attack.