Chemistry > Organic Chemistry > Amines
Read the passage and answer:
Amines have a lone pair of electrons on nitrogen atom due to which they behave as Lewis base. Greater the value of K or smaller the value of pK, stronger is the base. Amines are more basic than alcohols, ethers, esters, etc. The basic character of aliphatic amines should increase with the increase of alkyl substitution. But it does not occur in a regular manner as a secondary aliphatic amine is unexpectedly more basic than a tertiary amine in aqueous solutions. Aromatic amines are weaker bases than ammonia and aliphatic amines. Electron releasing groups such as –CH₃, –OCH₃, –NH₂ etc., increase the basicity while electron-withdrawing substituents such as –NO₂, –CN, halogens etc., decrease the basicity of amines.
(a) Arrange the following in the increasing order of their basic character. Give reason:
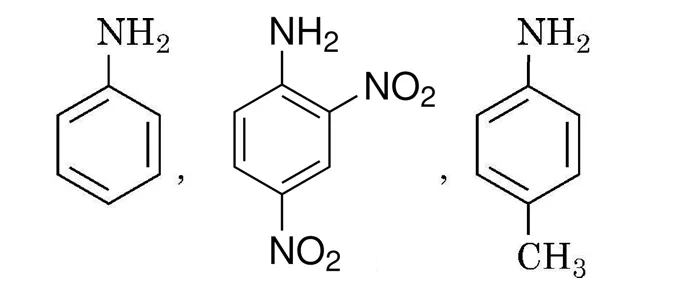
(b) Why is pK of aniline more than that of methylamine?
Keep Practicing
Edvaya Target
Board & Competitive Exams Made Easy
Edvaya Target is built for students preparing for JEE (Main & Advanced), NEET UG, and Board Exams (CBSE, ISC & State Boards) — using a practice-first approach that actually works.
Current Question
Community Stats
Track Your Progress
Sign up to save your practice progress, add favorites, and get personalized insights.

